
Distribution & Delivery

Scripts at the speed of now
Cost Containment

Control costs without affecting care
White-glove Service

Accredited, exceptional support
Compliance & Security

Fully prepared, fully protected
Data Management & Reporting

All pharmacy needs, one central dashboard
Dispensing Tech & Equipment

Reliable dispensing, flexible packaging
GETTING STARTED
Overview
Seamless Transitions

Effortless, 3-week changeover
See all Pharmacy Services

Quick view of our key services












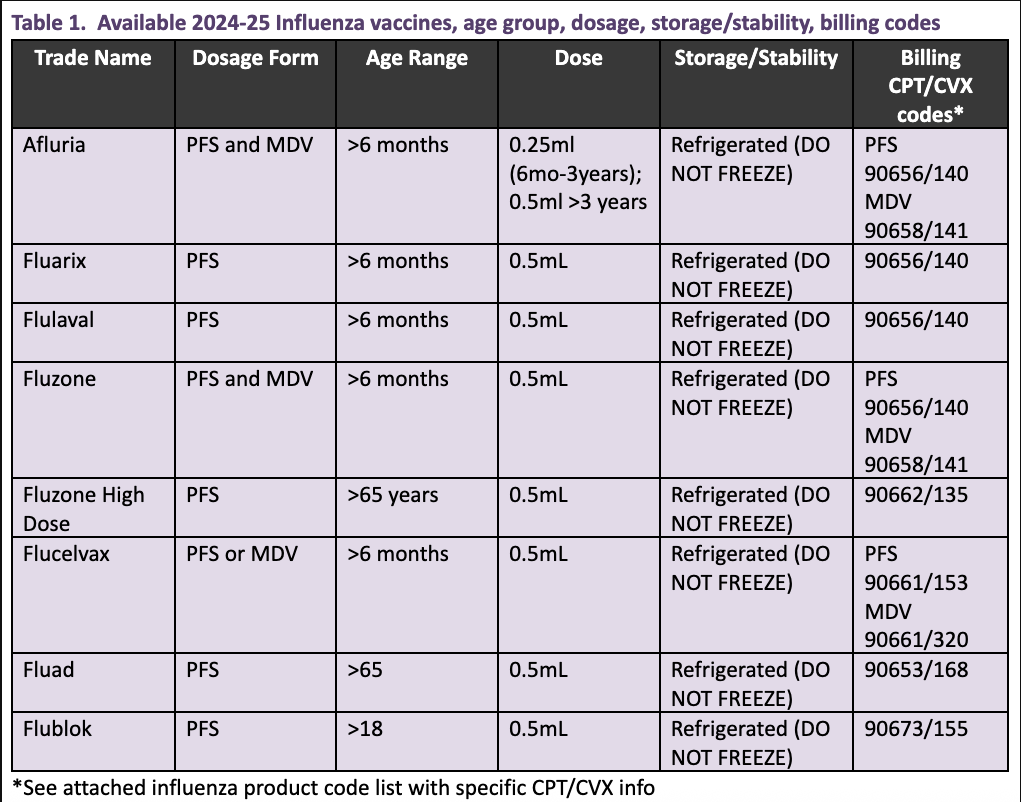
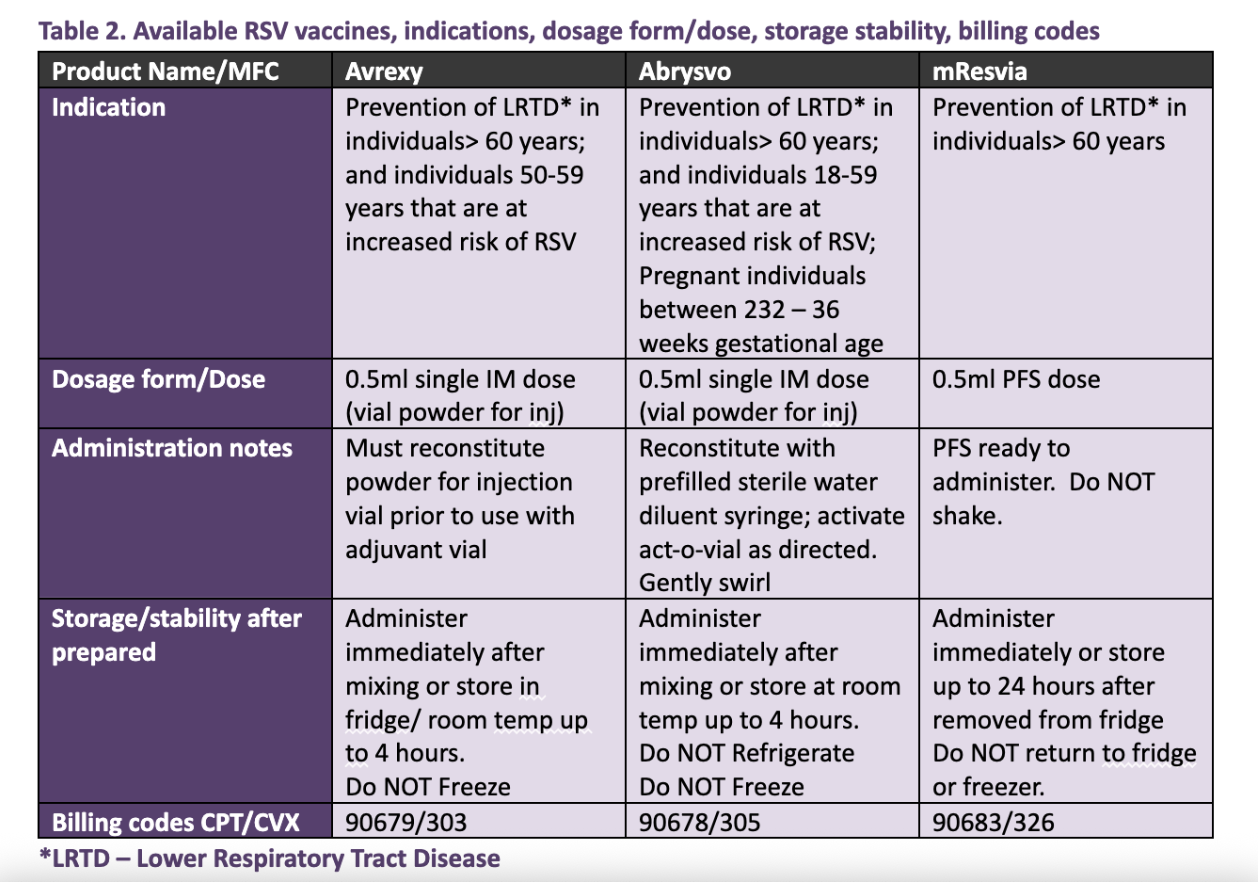
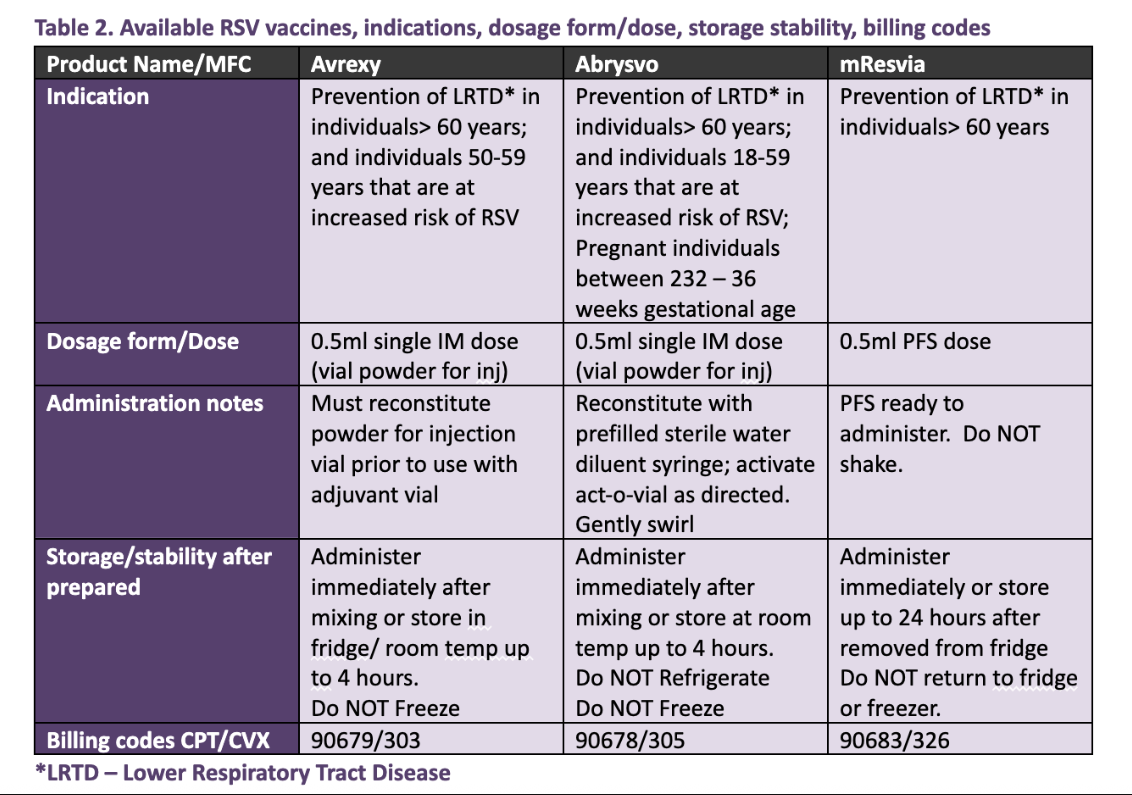


.jpg)